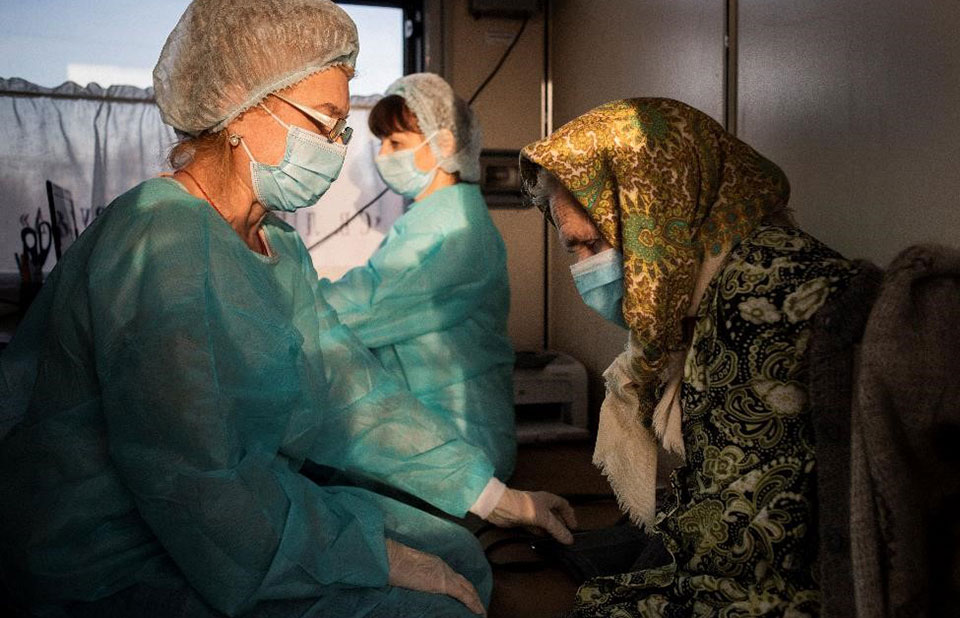
The global AIDS response has lost a great champion
UNAIDS is deeply saddened at the passing of Archbishop Desmond Tutu who fought against apartheid in South Africa and combated racism and injustice worldwide. He was a powerful voice in the fight against AIDS, combating denial, demanding access to treatment for all, calling out against discrimination of people living with...